
Can a small chamber placed on bare soil tell you whether a forest is absorbing carbon or releasing it?
That question drove me to spend an entire growing season on my knees in experimental birch plots, pressing a LICOR 6400-09 soil respiration chamber against the ground and watching CO₂ readings appear in real time. Month after month. Plot after plot.
What I measured surprised me. And it changed how I think about soil carbon monitoring tools entirely.
If you work in forest ecology, soil science, carbon cycle research, or environmental monitoring, this article will show you exactly how the LICOR soil respiration chamber works, when to use it, what it actually tells you, and why it remains one of the most trusted field tools in terrestrial carbon research.

What Is a Soil Respiration Chamber?
A soil respiration chamber is a field instrument that measures the rate of CO₂ released from the soil surface, a process called soil CO₂ efflux or soil respiration.
Soil respiration is the combined result of two biological processes happening underground at the same time. Root respiration releases CO₂ as plant roots consume oxygen and metabolise sugars. Microbial respiration releases CO₂ as soil microbes break down organic matter. Together these two processes make soil one of the largest sources of CO₂ flux in terrestrial ecosystems, releasing an estimated 60 billion tonnes of carbon per year globally.
Measuring that flux accurately in real field conditions is not simple. That is where the LICOR system comes in.
How Does the LICOR 6400-09 Soil CO₂ Flux Chamber Work?
The LICOR 6400-09 is a closed dynamic chamber system. Here is how it works step by step.
A cylindrical soil collar is inserted into the ground a few days before measurement. This collar stays in place throughout the measurement campaign so the soil disturbance from insertion has time to settle before readings begin. On measurement day, the chamber head is placed directly on top of the collar, forming an airtight seal.
Inside the chamber, air circulates continuously between the chamber and the LICOR 6400 infrared gas analyser. The analyser measures CO₂ concentration using infrared absorption, one of the most accurate methods available for gas analysis. As soil CO₂ diffuses upward and accumulates inside the sealed chamber, the analyser tracks the rate of concentration increase over time.
That rate of increase, combined with the chamber volume and soil surface area, gives you the CO₂ efflux in micromoles per square metre per second. The system also records soil temperature and soil moisture simultaneously, which is essential for interpreting what drives the flux.
In my field experiment, I placed collars in every experimental subplot and measured CO₂ efflux monthly from June through to late August. Each measurement took around two minutes per collar. I recorded soil temperature at 2 cm depth alongside each flux reading, which later allowed me to model how temperature responses differed between tree genotypes and treatments.
What Does Soil CO₂ Efflux Actually Tell You?
This is the question most people get wrong when they first start using soil respiration chambers.
CO₂ efflux is not the same as carbon loss. It is a rate measurement, a snapshot of how fast carbon is currently moving from soil to atmosphere at the moment of measurement. To understand whether a soil is gaining or losing carbon overall, you need to compare that efflux rate against the carbon inputs coming in from plant roots and leaf litter.
What efflux does tell you very clearly is how active the soil biological system is at any given time and under any given set of conditions. In my research, warmed plots showed 24% higher efflux in one birch genotype and 36% higher efflux in another compared to unwarmed controls. That told me directly that even a 0.9°C temperature increase was accelerating microbial and root activity in the soil, pushing more stored carbon into the atmosphere.
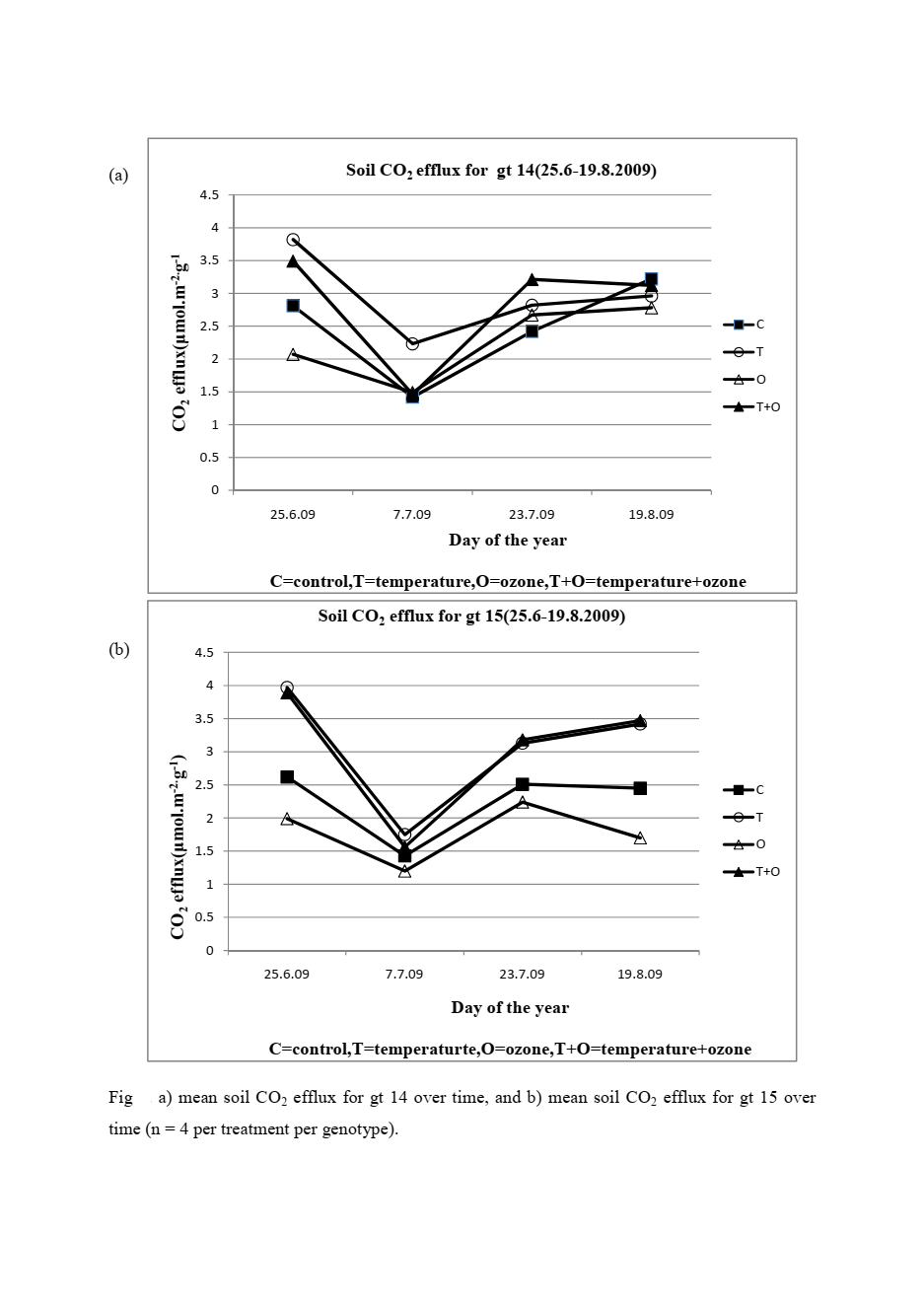
That finding was not just a number on a spreadsheet. It was evidence that small warming shifts can measurably alter the soil carbon balance, which has real implications for how we model carbon stocks under climate change scenarios.
Key Specifications: LICOR 6400-09 Soil Chamber
For researchers choosing equipment, here are the main technical details worth knowing.
The LICOR 6400-09 connects directly to the LI-6400XT portable photosynthesis system, making it a flexible attachment rather than a standalone instrument. The chamber volume is approximately 991 cm³ and the soil collar area is 71.6 cm². The infrared CO₂ analyser operates in the 0 to 3000 ppm range with a resolution of 0.1 ppm, giving it sensitivity that is more than sufficient for typical soil efflux rates. The system measures soil temperature using a thermocouple probe inserted alongside the collar at shallow depth.
One practical detail I learned quickly in the field: collar insertion depth affects your readings. Inserting the collar too deep cuts roots and triggers a temporary CO₂ pulse that biases measurements upward. Inserting it too shallow risks poor sealing and underestimation. The standard recommendation is 1 to 2 cm depth, and waiting at least 24 to 48 hours after insertion before measuring.
When Should You Use a Soil Respiration Chamber?
The LICOR soil respiration chamber works best in specific research contexts.
Use it for carbon flux studies where you need precise, replicated measurements across multiple plots or treatments. It is ideal for climate change experiments where you are comparing warmed versus control plots, elevated CO₂ versus ambient, or different vegetation types. It works well for seasonal monitoring studies where you need to track how efflux changes through a growing season.
It is less suitable for very large-scale spatial surveys where the measurement time per point becomes impractical, or for waterlogged soils where collar sealing is compromised. For broad landscape-scale carbon flux monitoring, eddy covariance tower systems complement chamber measurements by capturing ecosystem-scale fluxes that individual chambers cannot.
In my research, the chamber approach was ideal precisely because I needed to compare individual subplot responses to different treatments across a controlled experiment. The spatial resolution and treatment-level precision that chambers provide cannot be replicated by tower systems at that scale.
LICOR vs Other Soil Respiration Systems
Several other systems compete in this space and it is worth understanding the differences.
The EGM-5 from PP Systems is a popular alternative, also using infrared gas analysis with a closed chamber approach. It is generally considered more affordable and portable, making it attractive for researchers with budget constraints or high mobility needs. The trade-off is typically in measurement precision and data logging capability compared to the LICOR system.
The LI-8100A from LICOR itself is the newer dedicated soil flux system designed specifically for long-term automated monitoring. It supports multiplexed chamber arrays that can cycle through multiple collars automatically, making it far more suited to high-frequency unattended monitoring than the 6400-09, which requires manual operation.
For manual, high-precision field campaigns with treatment comparisons, the 6400-09 system I used remains a strong choice. For automated long-term monitoring stations, the LI-8100A is the more appropriate tool.
What Affects Soil CO₂ Efflux Readings?
Understanding the variables that drive flux is as important as the measurement itself.
Soil temperature is the strongest driver of short-term variation. Warmer soils respire faster, following a relationship described by the Q10 coefficient, typically between 1.5 and 3.5 for most soils. In my measurements, temperature differences between warmed and control plots explained most of the treatment effects I observed.
Soil moisture has a strong nonlinear effect. Very dry soils show suppressed respiration because microbial activity slows without water. Very wet soils also show reduced aerobic respiration as oxygen becomes limiting. Peak efflux typically occurs at intermediate moisture levels. Recording soil moisture alongside every flux measurement is not optional. It is essential for interpreting what you are seeing.
Root activity varies seasonally and with tree phenology. In my birch experiment, efflux rates peaked in mid-summer when root growth and leaf area were at their maximum, then declined toward late August as the season progressed. This seasonal pattern was consistent across genotypes but differed in magnitude between them.
Start Measuring Soil Carbon Flux With Confidence
If you are building a field monitoring setup for soil carbon research, forest ecology, or climate change experiments, the LICOR 6400-09 gives you the measurement precision and flexibility to capture real treatment effects in real conditions.
I used it across a full growing season in a controlled field experiment and the data it produced directly supported the key findings of my research. The instrument is reliable, well-documented, and supported by a broad scientific literature that makes your results directly comparable to studies worldwide.
Before you start, insert your collars at least 48 hours in advance, always record soil temperature and moisture alongside efflux, and plan your measurement schedule around consistent time-of-day windows to reduce diurnal variation in your dataset.
If you want to go deeper on the soil carbon monitoring tools used in environmental field research, explore the full Soil and Carbon Systems Tools section of this site. Every tool covered comes from real research application, not product catalogues.
Frequently Asked Questions
What is the difference between soil respiration and soil CO₂ efflux?
They refer to the same process from different angles. Soil respiration is the biological activity, the combined root and microbial metabolism that produces CO₂ underground. Soil CO₂ efflux is the physical measurement of that CO₂ as it exits the soil surface. In practice, researchers use both terms to describe what chamber instruments measure.
How often should soil respiration be measured in a field experiment?
It depends on your research question. For seasonal studies, monthly measurements capture the main temporal trends without excessive effort. For short-term warming or drought experiments where conditions change rapidly, weekly or even daily measurements give better resolution. In my experiment, monthly measurements from June to August captured the key seasonal pattern and treatment differences clearly.
Does soil collar insertion depth affect CO₂ flux readings?
Yes, significantly. Deep insertion cuts more roots and stimulates a temporary CO₂ pulse from wound respiration. Shallow insertion risks poor sealing and gas leaks. The standard depth of 1 to 2 cm is a practical balance, and the 24 to 48 hour settling period before measurement is not optional if you want reliable baseline readings.
Can the LICOR 6400-09 measure soil respiration in waterlogged soils?
It is not ideal for waterlogged conditions. Water in the collar can interfere with the chamber seal and the CO₂ signal. For wetland or peatland measurements, specialised floating chamber systems or purpose-built wetland flux chambers are more appropriate.
What is a typical soil CO₂ efflux rate in a temperate forest?
Rates vary widely depending on temperature, moisture, vegetation, and season. Typical values in temperate and boreal forests range from around 1 to 6 micromoles of CO₂ per square metre per second during the growing season. In my warmed experimental plots, efflux consistently exceeded control plots across all measurement dates, with the largest differences observed in mid-summer at peak soil temperature.
How do you calculate annual soil carbon efflux from chamber measurements?
You interpolate between measurement dates, typically fitting a temperature-response function to your data using Q10 or Arrhenius-type models, then integrate over the year using continuous soil temperature records. This approach converts discrete chamber measurements into an annual carbon flux estimate, which can then be compared against carbon input estimates to assess whether the soil is a net sink or source.








