
There is a simple question that keeps resurfacing whenever we look at wood not as material, but as stored energy:
Why is it that some biological carbon breaks down within years, while other forms seem to remain locked away for decades or even centuries?
That question becomes very concrete when you start working with actual biomass systems. Wood is everywhere, but turning it into fuel is anything but straightforward.
Unlike first-generation biofuels that rely on food crops like corn or sugarcane, lignocellulosic ethanol is built from what we usually treat as waste, wood chips, crop residues, and fast-growing grasses. In theory, the feedstock is abundant enough to reshape energy systems. In practice, the chemistry resists.
The problem is not availability. It is structure.
The hidden architecture inside wood
At a glance, wood looks simple. But under the surface, it behaves more like a engineered composite than a natural material.
Cellulose forms long chains of glucose that could, in principle, be fermented into ethanol. But those chains are tightly packed into crystalline regions that resist breakdown.
Hemicellulose surrounds them like a flexible matrix, chemically more diverse and easier to hydrolyze, but unpredictable in fermentation pathways.
Then there is lignin, the part that changes everything. A dense aromatic polymer, lignin essentially acts like a biological plastic. It evolved to protect plants from decay, and it does that job extremely well. From a biofuel perspective, it becomes the main barrier.
When structure becomes a processing problem
To access the sugars locked inside wood, the material has to be pre-treated before anything biological can happen.
This is where the system starts to become industrial rather than biological.
Steam explosion physically tears the structure apart using pressure and heat. Acid hydrolysis dissolves hemicellulose and exposes cellulose, but at chemical cost. Ionic liquids offer a more precise molecular approach, but remain expensive at scale.
Even after that, enzymes are introduced, cellulases that act like molecular scissors. But the system is fragile. As sugars begin to accumulate, they can inhibit the very enzymes meant to produce them. The process slows down just when it should accelerate.
Why this doesn’t scale easily
On paper, the chemistry works. In reality, economics dominates.
Pre-treatment demands energy and infrastructure. Enzymes are sensitive and costly. And lignin, the most abundant structural component, remains underused, often just burned to keep the system running.
What this really reveals is that we are not just dealing with a chemical conversion problem. We are dealing with a system design problem.
Why biological systems still matter here
In long-term field studies on forest systems, small changes in temperature alone can shift carbon dynamics in ways that are surprisingly large.
In my silver birch field experiments, where I collected and analyzed soil respiration data, I measured that a temperature increase of less than one degree Celsius raised soil CO₂ efflux by up to 36% depending on genotype. That kind of response highlights something important: carbon stored in biomass is never isolated from the biological systems around it.
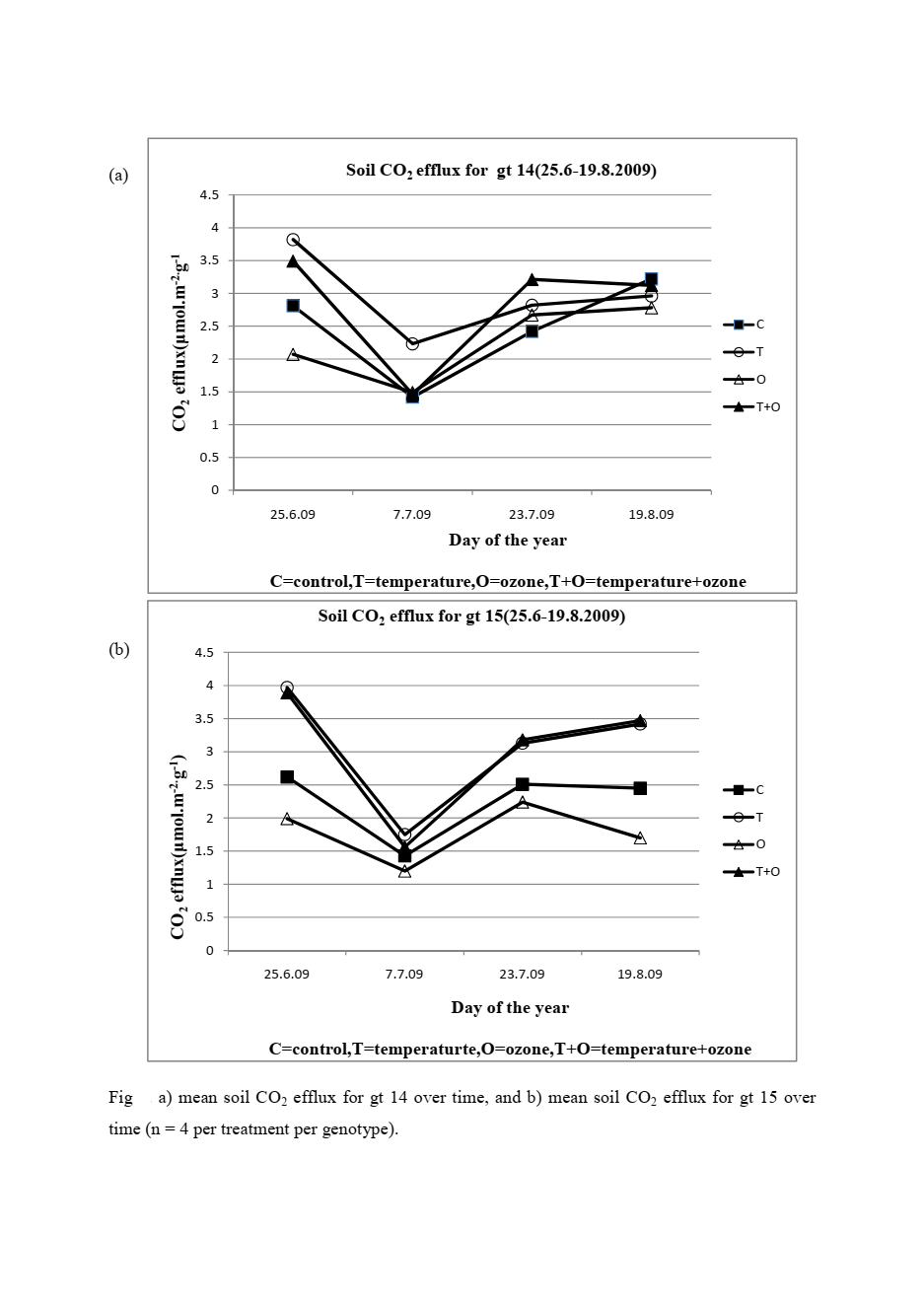
Roots, microbes, and soil respiration all interact with that carbon continuously. So when we try to extract energy from woody biomass at scale, we are not just processing material, we are intervening in an active carbon cycle.
That interaction is often where models oversimplify reality.
The key limiting factor that consistently emerges is lignin
If cellulose is the fuel potential, lignin is the constraint.
It cannot be fermented. It resists enzymatic attack. And yet it represents a large fraction of the biomass.
Until lignin can be efficiently converted into value, whether chemicals, materials, or higher-grade fuels the system remains economically limited.
This is why “lignin valorisation” has become one of the central research directions in bioenergy. It is not just waste management. It is the key to viability.
Two ways forward: biology and thermochemistry
There are two dominant routes being developed in parallel.
The biochemical route relies on enzymes and fermentation, closer to natural processes, but slow and sensitive.
The thermochemical route uses gasification, converting wood into syngas (CO and hydrogen) under low oxygen conditions. From there, liquid fuels such as sustainable aviation fuel can be synthesized.
One is biological precision. The other is chemical force. Most future systems will likely combine both.
Conclusion
Lignocellulosic ethanol is often described as a fuel of the future. But the real question is not whether wood contains energy. It clearly does.
The question is whether we can break down a material that evolved specifically to resist breakdown, in a way that is both energy-efficient and economically viable.
Wood is not refusing to yield energy. It is simply doing what it was designed to do, stay intact.
FAQs
What makes lignocellulosic ethanol different from regular ethanol?
Regular ethanol comes from easily accessible sugars in crops like corn or sugarcane. Lignocellulosic ethanol is made from woody biomass, where sugars are locked inside complex structures like cellulose and lignin, making extraction far more difficult.
Why is lignin such a major limiting factor?
Lignin acts as a rigid protective barrier around cellulose. It resists chemical and biological breakdown, and until it is disrupted, enzymes cannot efficiently access the fermentable sugars.
Why isn’t wood-based ethanol already widely used if biomass is abundant?
The challenge isn’t supply, it’s processing. Breaking down wood requires energy-intensive pre-treatment and costly enzymes, which currently makes production less competitive than fossil fuels.
Is the process energy efficient overall?
It can be, but efficiency depends on how well heat, chemicals, and byproducts are managed. Poorly optimized systems can consume a large share of the energy they produce.
What happens to lignin after processing?
Most facilities burn lignin to generate heat and power. While practical, this is a low-value use, which is why research is focusing on converting lignin into higher-value materials.
Are there alternatives to fermentation for converting wood into fuel?
Yes. Gasification converts biomass into syngas, which can then be processed into liquid fuels like synthetic diesel or aviation fuel.
How does real field research connect to these systems?
In my field experiments on silver birch, even small warming increased soil CO₂ flux, showing how sensitive carbon systems are. That same sensitivity helps explain why unlocking carbon from wood is not just a chemical challenge, but a system-level one.
What needs to improve for this technology to scale?
More efficient pre-treatment methods, lower-cost enzymes, and better use of lignin are key. Progress is steady, but scaling remains the main barrier.









